Partnerships
As regulatory science is best served by large and translational projects as well as open lines of communication between various stakeholders, the PRI strives to be the nexus between academia, industry, and regulators. For this purpose, the PRI works actively on, i) identifying important microbiome regulatory science topics, ii) defining strategies to address these topics, iii) participating in initiatives aimed at consensus building, and iv) promoting the emergence of various pre-competitive projects leading to emergence of new standards and tools.
Today the PRI is working through partnerships and collaborations to develop a thorough, robust, and appropriate knowledge and therefore best represent all stakeholders involved in microbiome-based medicinal products development when legislations and regulatory frameworks are being updated.

Aligning the future of human microbiome research and standardisation in Europe

As a partner within the Human Microbiome Action project, the PRI is co-lead of the Ethics and Regulatory work package. As such, the PRI will aim at formalizing a consensus emerging from the various work packages into regulatory-sound proposals aligned with regulators’ needs and taking into account the current legislative developments at the EU level (Blood tissue and cells legislation as well as the general pharmaceutical legislation revision).
The PRI will also be involved in the drafting of a roadmap for the implementation of an EU microbiome-dedicated surveillance network aiming at ensuring the emergence of standards, harmonization and updating, for the years to come.
Find out more about the Human Microbiome Action Project | EU Project N° 964590
Understanding the microbiome has enormous potential in public health, particularly in the prevention and treatment of many non-communicable diseases. Yet despite promising advances in microbiome research in recent years, meaningful clinical applications, scientifically-sound diet and lifestyle recommendations and public health guidance from research outcomes remain elusive. One crucial reason for this shortfall in harnessing the microbiome’s potential is the lack of commonly used and accepted foundational approaches within research.
The Human Microbiome Action project will develop a range of guidance documents and recommendations for researchers, public health officials and industry stakeholders to provide roadmaps for necessary foundations and key actions that allow for aligned microbiome research in the future.
Microbiome regulatory science across food systems and the environment

Within MicrobiomeSupport, the PRI has taken part in the “MicrobiomeSupport Regulatory Workshop” where it made a presentation on the importance of developing awareness in microbiome regulatory science from academia to industry, all the way to regulators.
Based on feedback obtained from various stakeholders within the context of the project and their integration about the main challenges encountered, it appeared that regulatory questions were at the heart of their concerns. In this workshop, the PRI proposed some tools which could be used to improve regulatory awareness and introduce regulatory science activities with the objective of optimizing translation from academia to industry.
Find out more about the MicrobiomeSupport Project | EU Project N° 818116
MicrobiomeSupport will support the need for a better alignment of R&I (policy) agendas in Europe and worldwide. The improvement of the following 5 key policy challenges can lead to innovative applications in the promising microbiome field, according to a September 2017 OECD-Report.
- Science policy
- Enabling translational science
- Public-private collaboration
- Regulatory frameworks
- Skills, communication and the public
The MicrobiomeSupport project will address these as well as additional points to enable innovative microbiome applications in the food system and beyond generating true benefit for our society.
Integrating Microbiome Drug Metabolism in Pharmaceutical Development – MEDICEN Microbiome Drug Metabolism Working Group

Within this expert group, the PRI provides microbiome regulatory science input and introduces regulatory mindsets into the discussions in order to translate consensus and new approaches emerging from complementary expertise into regulatory-sound proposals. Through its experience with regulators’ understanding of the microbiome, the PRI points out the necessary documentation in order to “educate” regulators as well as various stakeholders involved in drug development and for whom the microbiome is a novel area. In collaboration with Medicen, the PRI is participating in the identification of Microbiome regulatory science research topics which could lead to the emergence of pre-competitive projects investigating the field of pharmacomicrobiomics.
The Medicen cluster, based in France, federates a large network of excellence in innovation and brings together private and public actors around the challenges associated with the development of tomorrow’s medicinal products.
In the area of microbiome, Medicen has initiated an expert working group on the topic of drug-induced and drug-related changes in microbiome.
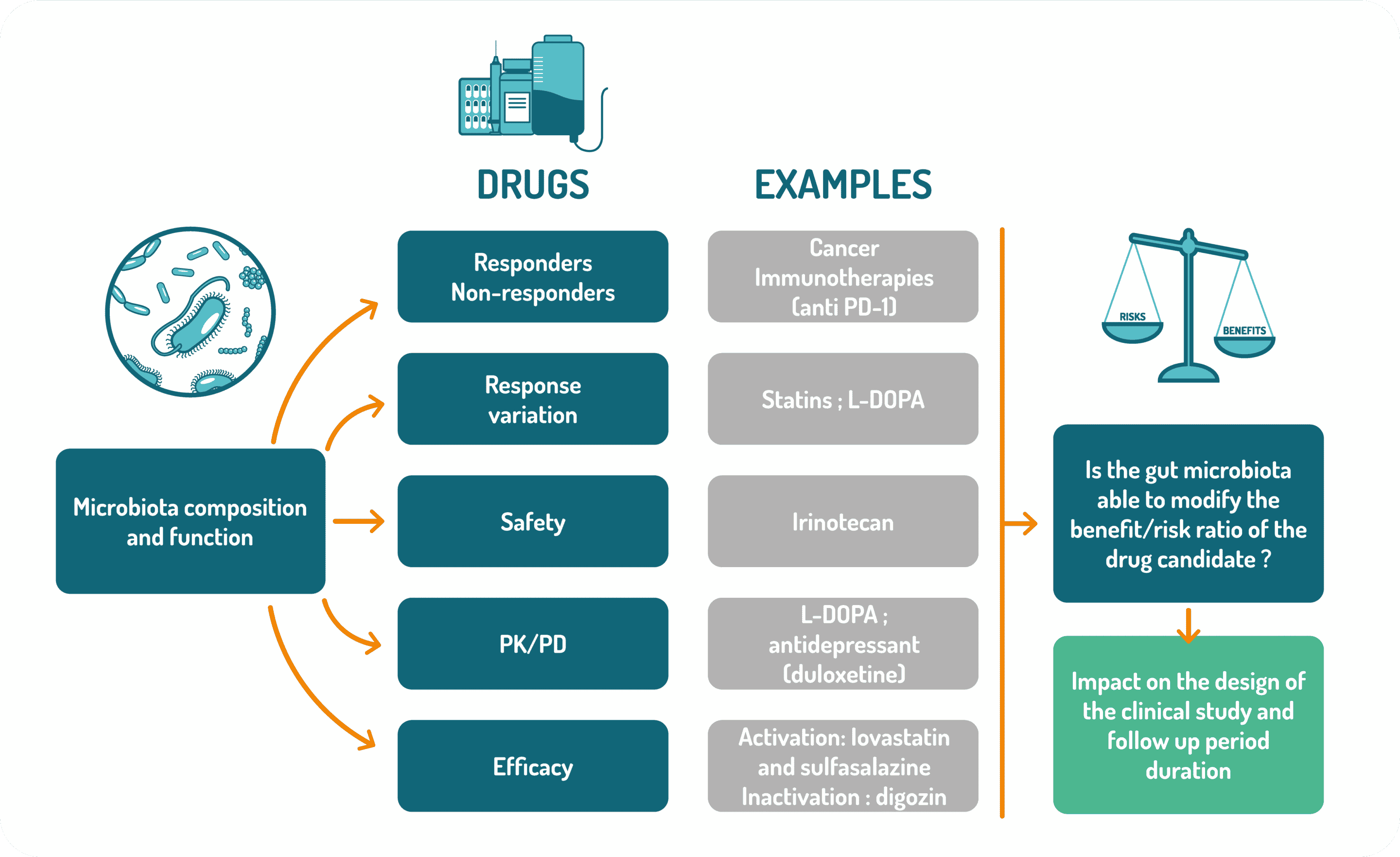
Pharmacomicrobiomics is an emerging field that investigates the interplay of microbiome variation and drug response and disposition (absorption, distribution, metabolism and excretion). This illustration shows examples of the potential impacts of the gut microbiota composition/function on drug efficacy, safety and PK/PD. Based on these potential impacts of the gut microbiota on drug response and disposition, the MEDICEN working group is analyzing how it could be useful to include the gut microbiota’s impact on the risk/benefit ratio assessment during drug development, not only in preclinical but also in clinical studies.